by
Olga Deshchenko, DOTmed News Reporter | October 07, 2010
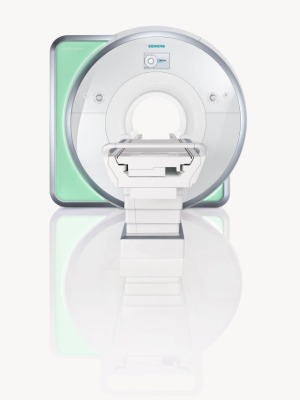
Siemens Healthcare
Magnetom Skyra 3T MRI
The Magnetom Aera 1.5 Tesla and the Skyra 3 Tesla MRI scanners from Siemens Healthcare received U.S. Food and Drug Administration approval and are now commercially available in the United States, the company announced Thursday.
The 70-centimeter open bore systems come equipped with Tim 4G, a technology that starts with 48 radio frequency channels at both 1.5T and 3T, "presently the highest standard RF channel configuration ever offered at either field strength," according to the release.
"Siemens is committed to help address the challenges in the health care industry," said Don Fowler, the company's VP, magnetic resonance, in prepared remarks.



Ad Statistics
Times Displayed: 348574
Times Visited: 21068 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
The systems also come with the Dot (Day optimizing throughout) engine, which enables clinicians to customize frameworks for individual patients and offers exam automation options to enhance the MR workflow.
Siemens service and field technicians will learn the ins and outs of the new Magnetom Skyra 3T MRI at the company's Cary, N.C.-based training center, according to
Corey Hess, sales director of National MRI Shielding. The Dallas, Texas-based company provided the RF shielding enclosure for the unit at the center.
The Magnetom Aera and Skyra systems have been commercially available outside the United States since Nov. 2009. The company said it has sold more than 100 systems to date.
