by
Lauren Dubinsky, Senior Reporter | October 06, 2016
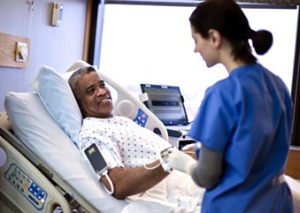
Can be used with the
Philips Wearable Biosensor
Royal Philips has launched the next generation of its IntelliVue Guardian software for use with the Philips Wearable Biosensor patient monitoring device.
The biosensor is a medical-grade, self-adhesive, single-use, wireless device that continuously measures heart rate, respiratory rate, posture, and detects falls while being worn on the patient's chest. That data is then sent to the IntelliVue Guardian software, which analyzes the measurements over a period of time to spot trends and then notifies the caregiver or clinician if the preset limits are exceeded.
"This combined solution provides caregivers with peace of mind that their patients are being monitored frequently for signs of deterioration between spot checks while they are attending to other tasks seamlessly in their usual workflow," Carla Kriwet, business leader of patient care and monitoring solutions at Philips, told HCB News.



Ad Statistics
Times Displayed: 57652
Times Visited: 1710 Ampronix, a Top Master Distributor for Sony Medical, provides Sales, Service & Exchanges for Sony Surgical Displays, Printers, & More. Rely on Us for Expert Support Tailored to Your Needs. Email info@ampronix.com or Call 949-273-8000 for Premier Pricing.
The increasing rate of individuals with chronic diseases is contributing to 86 percent of health care costs, according to the Centers for Disease Control. Health care stakeholders are searching for cost-effective ways to monitor, diagnose and treat patients.
"The IntelliVue Guardian and Philips Wearable Biosensor solution can now create actionable information and insight to have a very positive impact on the chronically ill by helping to reduce associated costly adverse events, complications, unplanned transfers back to the ICU and longer lengths of hospitalization," said Kriwet.
The new iteration of the software is configurable with Early Warning Scoring protocols at most institutions and offers new features like the SureSigns VS4 spot check monitors and the IntelliVue MP5SC monitor.
The software is available in countries requiring CE mark and was showcased at the 29th European Society of Intensive Care Medicine Annual Congress in Milan this past week.
Both the software and the biosensor are 510(k)-pending and not currently for sale in the U.S.

