by
John R. Fischer, Senior Reporter | October 23, 2017
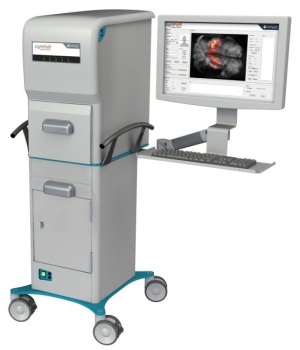
The LightPath Imaging System will be part of
a project for assessing Cerenkov Luminescence
Imaging technology in identifying cancerous
tissue during prostate cancer surgery
Identifying cancerous tissue during prostate cancer surgery may soon be an easier feat for surgeons, through the use of a new molecular imaging technology currently under evaluation.
Lightpoint Medical is teaming up with Royal Philips, the Netherlands Cancer Institute and the University of Twente to assess and compare the use of Cerenkov Luminescence Imaging to pathology for identifying cancerous tissues during procedures and ensuring more accurate removal of them.
“Surgeons do not have any accurate tools for detecting prostate cancer during surgery,” David Tuch, the CEO of Lightpoint Medical, told HCB News. “Frozen section pathology is used in a small handful of centers but is not used more widely because it’s slow, expensive and requires specialized staff. Consequently, there is a high medical need for accurate tools to detect cancer during prostate cancer surgery.”



Ad Statistics
Times Displayed: 6368
Times Visited: 20 Brand-New FDA-cleared Advanced Ultrasound Medical Device available for sale or lease to Wound Care Centers or any other Medical Facilities.The Arobella 1000D is designed for non-contact or debridement ultrasound wound healing therapy, or any other wounds
Completely removing prostate cancer during surgery can be challenging due to the difficulty in distinguishing healthy tissue from cancerous, especially when the disease is advanced. The decision to examine Cerenkov Luminescence Imaging follows a recent scientific discovery that PET imaging agents emit light.
The four, in a joint research project, will evaluate the technology’s ability to image the biomarker, Prostate Specific Membrane Antigen (PSMA). It will include the use of Lightpoint Medical’s CE-marked LightPath Imaging System, which uses Cerenkov Luminescence Imaging technology to acquire molecular images of tissue specimens, as well as Philips' Vereos digital PET/CT system, providing Lightpoint Medical with the opportunity to connect its intra-operative technology with Philips’ preoperative PET imaging.
Should the study prove successful, larger clinical trials will take place with the aim to spare healthy tissue and reduce recurrence rates in prostate cancer.
Other innovations for the treatment of prostate cancer are currently undergoing testing, including a new
targeted alpha therapy protocol developed by German researchers, as well as a new
radiotracer that takes minutes to manufacture and was developed by British researchers.
Tuch says the collaboration brings together some of the greatest scientific minds and could assist in introducing a more accurate tool for ensuring success in prostate cancer surgery.
“We were foremost attracted by the very high clinical and scientific calibre of the collaborators,” he said. “Additionally, this was an opportunity to evaluate a prostate cancer imaging agent targeting Prostate Specific Membrane Antigen (PSMA). PSMA imaging has been a major advance in diagnostic and follow-up imaging but has not yet been applied widely yet for surgical guidance.”
The Dutch Cancer Society has contributed funding to the project, which is currently underway.

