by
John R. Fischer, Senior Reporter | December 21, 2020
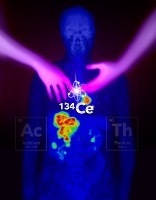
Cerium-134 enables clinicians to visualize the therapeutic effects of actinium and thorium on cancer in the body through PET imaging
Scientists in the U.S. Department of Energy (DOE) Isotope Program have developed Cerium-134, a radionuclide that enables PET imaging of actinium and thorium, alpha emitters used for cancer therapy.
"When administered prior, Ce-134 imaging capability will show if the actinium-225 and thorium-227 treatment has potential to work. When administered after, Ce-134 will show if the actinium-225 and thorium-227 treatment did work, and to what extent. It is critical for developing the medication. It is critical for evaluating the actinium-225 and thorium-227 medication on a case-by-case basis," Dr. Stosh Kozimor, lead Los Alamos National Laboratory researcher on the joint project with Lawrence Berkeley National Laboratory, the University of California, and the University of Wisconsin, told HCB News.
Actinium and thorium radionuclides can be used in targeted alpha therapies to treat cancer, with the potential to spare patients from negative side effects.



Ad Statistics
Times Displayed: 123266
Times Visited: 7153 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
Cerium-134 is produced with a linear accelerator and attaches to the same chelate/ligant as actinium-225 and thorium-227. It emits a positron that is picked up by the PET scanner. Users can use this to then track the therapeutic and identify its proximity to the diseased tissue being examined.
Large-scale production and purification methods were developed to overcome the incompatibility of
actinium-225 and thorium-227 to PET imaging, which is essential to studying their therapeutic effect in the body, according to Kozimor.
The demonstration is necessary in helping to make the isotope available in clinical settings.
Funding was provided by the DOE Isotope Program, the DOE, a DOE Integrated University Program graduate research fellowship and a Nuclear Regulatory Commission Faculty Development Grant.
Cerium-134, actinium-225 and thorium-229 are available from the DOE Isotope program.
"The DOE Isotope Program is working to increase routine availability of the cerium-134 isotope," said Kozimor. "The isotope will be incorporated into radiopharmaceuticals that include the biological targeting mechanism. These will be tested, and then submitted to the FDA for approval."

