by
Brendon Nafziger, DOTmed News Associate Editor | October 02, 2009
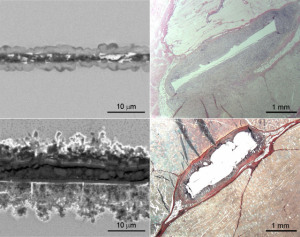
The mangesium-zinc-calcium
glass sheds no bone-growth-
dampening gas bubbles (top images),
while normal magnesium alloys (below)
are rich with harmful
hydrogen activity
Scientists in Switzerland have developed a type of biodegradable metallic glass that could one day replace titanium and stainless steel as the metal that bone screws and other surgical implants are made of, according to an article in Nature Materials published online this week.
Because the new material created by scientists at ETH Zurich in Switzerland breaks down gradually and safely in the body, it could be used to manufacture orthopedic implants, such as bone screws -- tools that hold bones in place so they can mend -- which won't require follow-up surgeries for removal.
The magnesium hurdle



Ad Statistics
Times Displayed: 60894
Times Visited: 1960 Ampronix, a Top Master Distributor for Sony Medical, provides Sales, Service & Exchanges for Sony Surgical Displays, Printers, & More. Rely on Us for Expert Support Tailored to Your Needs. Email info@ampronix.com or Call 949-273-8000 for Premier Pricing.
When it comes to biodegradable metals tough and flexible enough to hold bones in place, iron- and magnesium-based alloys are the best candidates.
But "since iron degradation is slow, the first choice is magnesium," Jörg Löffler, Ph.D., professor of metal physics and technology at ETH Zurich, and the corresponding author of the paper, tells DOTmed News.
Not only does magnesium degrade faster, when it dissolves, it releases harmless ions well tolerated by the body, says Dr. Löffler.
But magnesium has one important drawback: when it dissipates in the body, it releases hydrogen gas, forming bubbles that are bad news for the bone. "They stay around the implant, and hinder bone growth and thus the healing process," says Dr. Löffler.
The solution hit upon by Dr. Löffler and his colleagues, including Bruno Zberg, his doctoral student and the first author on the study, was to create a "so-called magnesium-based metallic glass that doesn't show any clinically observable hydrogen production," he says.
Glass production
The "glass" is produced by rapidly cooling a melt of magnesium, zinc and calcium. The speed of the cooling prevents atoms from forming the crystalline structure common to most metals.
"Thanks to this procedure, we can add much more zinc to the magnesium than is possible with conventional alloys," Dr. Löffler says. By adding more than 28 percent of zinc atoms to the alloy, a zinc- and oxygen-rich passivating layer forms on the metal's surface, which fundamentally alters the corrosion behavior of the material.
"Above 28 atomic percent zinc, degradation is slower and modified, so that in clinical studies we do not see any observable hydrogen evolution. No gas bubbles form around the implant," he says.
Peaceful degradation
Key to the success of the implant would be how safe its decomposition is for the body. And here Dr. Löffler says the material really shines. The amount of magnesium released by a hypothetical implant made from this alloy would be orders of magnitude less than what most health agencies recommend people consume.
For example, the USDA recommends people get around 400 milligrams of magnesium a day, but a typical one-gram metallic-glass screw during its one-year lifespan might degrade out only 3 milligrams daily, less than a hundredth of the recommended amount of the metal.
Clinical possibilities
Currently, the Swiss team has only performed animal studies, where they found the metallic glass to be fully biocompatible -- and the surrounding tissue, hydrogen bubble-free.
Down the road, Dr. Löffler expects to run clinical trials on a few patients, "to be sure that all requirements are fulfilled for bone implant material," he says.

