by
Brendon Nafziger, DOTmed News Associate Editor | November 02, 2012
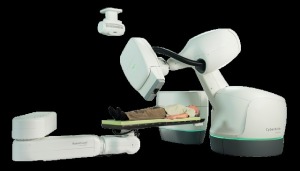
The CyberKnife M6 Series (Credit: Accuray)
Just days after its public debut at ASTRO 2012 in Boston, the next-generation CyberKnife M6 Series has received Food and Drug Administration clearance, the device's manufacturer Accuray Inc. has revealed.
Accuray said Thursday that the device and its new InCise Multileaf Collimator, available on the two higher-end models of the M6 Series, received FDA 510(k) clearance.
The device is designed for stereotactic radiosurgery and stereotactic body radiation therapy — both involve higher doses of radiation in fewer treatments than conventional radiation therapy.



Ad Statistics
Times Displayed: 348574
Times Visited: 21068 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
Accuray's new TomoTherapy line, which also debuted at ASTRO, had received FDA clearance before the show started, the company said.
The MLC is used to sculpt the X-ray beam to the size and shape of the tumor. It has a bigger opening than the CyberKnife's previous collimator and allows, theoretically, faster treatment times for some tumors, a company representative told DOTmed News this week. Researchers with Fox Chase Cancer Center in Philadelphia found theoretical treatments 35 to 70 percent quicker, he said.
The MLC allows the CyberKnife to better treat bigger, irregularly shaped tumors, the company said in its press release.
The new CyberKnife models are the FIM and FM, which feature the MLC, and the FI, which doesn't. The FM and FI are upgradeable.
The company said the equipment would probably start shipping, at least to Europe, later this year.
