by
Loren Bonner, DOTmed News Online Editor | April 04, 2014
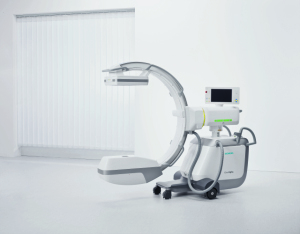
Siemens' Cios Alpha C-arm
Siemens Healthcare announced on Wednesday that the U.S. Food and Drug Administration has cleared its new mobile C-arm, the Cios Alpha.
The C-arm is a flat-panel detector that also combines a power output of 25 kilowatts and has a field of view that the company said is up to 25 percent larger than conventional C-arms.
Other features include workflow improvements such as a user-friendly touch screen interface, a unique position storage feature, and an active cooling system that helps to ensure exceptional image quality during prolonged interventions.



Ad Statistics
Times Displayed: 7059
Times Visited: 23 Brand-New FDA-cleared Advanced Ultrasound Medical Device available for sale or lease to Wound Care Centers or any other Medical Facilities.The Arobella 1000D is designed for non-contact or debridement ultrasound wound healing therapy, or any other wounds
"With imaging technology that helps overcome disruption and reduce inefficiency in the OR, the new Cios Alpha mobile C-arm will aid surgical teams in maximizing productivity and fostering a higher quality of patient care across multiple disciplines," said Laurie Fisher, vice president of X-ray products for Siemens Healthcare.
Siemens said the C-arm is ideal for vascular, orthopedic and urological procedures.
