Targeting estrogen and HER2 receptors through PET imaging
by Lauren Dubinsky, Senior Reporter | January 03, 2017
Molecular Imaging
Rad Oncology
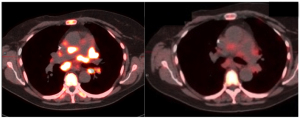
FDG-PET is currently one of the only molecular imaging techniques used in oncology — but that may soon change.
New PET probes being tested have shown promise, especially in breast cancer clinical trials. Experts at the Abramson Cancer Center and University of Pennsylvania believe that they can make it easier to diagnose, monitor and treat patients.
They published a review in JAMA Oncology asserting that finding a way to utilize these probes more widely in clinical settings should be a top priority.

 The two new classes of probes are designed to bind to estrogen and HER2 receptors. Breast, uterine and ovarian tumors typically use those receptors to grow faster, and because of that, many cancer drugs target them.
The two new classes of probes are designed to bind to estrogen and HER2 receptors. Breast, uterine and ovarian tumors typically use those receptors to grow faster, and because of that, many cancer drugs target them.
Detecting these receptors with PET scans allows oncologists to examine all sites of cancer on each patient. That will help them select the appropriate treatment more quickly and evaluate how successful a drug is at hitting its receptor targets.
These new probes can also uncover receptors or other tumor-related markers at sites where the cancer might have spread, including bone.
Probes that target breast cancer are showing the most promise in clinical trials. A national, multicenter trial co-chaired by an oncologist at the University of Pennsylvania focused on the estrogen receptor, and a smaller trial is underway at Abramson Cancer Center’s 2-PREVENT Translational Center of Excellence.
“Many of these methods are already being studied in clinical trials, but the path from clinical trials to routine clinical use is seldom easy,” Dr. David A. Mankoff of the university said in a statement. “And molecular imaging methods face some particularly challenging hurdles such as the need to deliver the short-lived imaging probes to centers performing the imaging.”
There is currently no standard approach for testing the probes because they are so new to oncologists. That makes it difficult to demonstrate their clinical value to the FDA, medical insurers and even oncologists.
Mankoff believes that the imaging and oncology fields need to come together to find the best way forward. He explained that in order to make a strong clinical case they’ll need to demonstrate the probes’ ability to produce better outcomes than traditional treatment.
In a recent trial, researchers showed that combining FDG with HER2-targeted PET imaging was 100 percent accurate at predicting patients’ responses to an expensive new anti-HER2 breast cancer drug. That helps to direct treatment to patients who are likely to benefit, and sparing others from side effects and the cost of ineffective drugs.
Most importantly, trials should focus on testing the new imaging methods on applications that it can perform better than other imaging or biopsy-based techniques, said Mankoff.
“These clinical trial results for the new molecular imaging methods are going to be compelling for patients and their referring oncologists only when they address clinical challenges not met by existing approaches,” he added.
New PET probes being tested have shown promise, especially in breast cancer clinical trials. Experts at the Abramson Cancer Center and University of Pennsylvania believe that they can make it easier to diagnose, monitor and treat patients.
They published a review in JAMA Oncology asserting that finding a way to utilize these probes more widely in clinical settings should be a top priority.
Your Trusted Source for Sony Medical Displays, Printers & More!
Ampronix, a Top Master Distributor for Sony Medical, provides Sales, Service & Exchanges for Sony Surgical Displays, Printers, & More. Rely on Us for Expert Support Tailored to Your Needs. Email info@ampronix.com or Call 949-273-8000 for Premier Pricing.

Detecting these receptors with PET scans allows oncologists to examine all sites of cancer on each patient. That will help them select the appropriate treatment more quickly and evaluate how successful a drug is at hitting its receptor targets.
These new probes can also uncover receptors or other tumor-related markers at sites where the cancer might have spread, including bone.
Probes that target breast cancer are showing the most promise in clinical trials. A national, multicenter trial co-chaired by an oncologist at the University of Pennsylvania focused on the estrogen receptor, and a smaller trial is underway at Abramson Cancer Center’s 2-PREVENT Translational Center of Excellence.
“Many of these methods are already being studied in clinical trials, but the path from clinical trials to routine clinical use is seldom easy,” Dr. David A. Mankoff of the university said in a statement. “And molecular imaging methods face some particularly challenging hurdles such as the need to deliver the short-lived imaging probes to centers performing the imaging.”
There is currently no standard approach for testing the probes because they are so new to oncologists. That makes it difficult to demonstrate their clinical value to the FDA, medical insurers and even oncologists.
Mankoff believes that the imaging and oncology fields need to come together to find the best way forward. He explained that in order to make a strong clinical case they’ll need to demonstrate the probes’ ability to produce better outcomes than traditional treatment.
In a recent trial, researchers showed that combining FDG with HER2-targeted PET imaging was 100 percent accurate at predicting patients’ responses to an expensive new anti-HER2 breast cancer drug. That helps to direct treatment to patients who are likely to benefit, and sparing others from side effects and the cost of ineffective drugs.
Most importantly, trials should focus on testing the new imaging methods on applications that it can perform better than other imaging or biopsy-based techniques, said Mankoff.
“These clinical trial results for the new molecular imaging methods are going to be compelling for patients and their referring oncologists only when they address clinical challenges not met by existing approaches,” he added.
You Must Be Logged In To Post A CommentRegisterRegistration is Free and Easy. Enjoy the benefits of The World's Leading New & Used Medical Equipment Marketplace. Register Now! |
|










