by
John R. Fischer, Senior Reporter | April 18, 2018
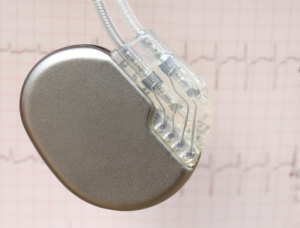
CMS has expanded coverage for
MR exams to Medicare patients with
certain implants
A greater number of Medicare beneficiaries with intact cardiac implants will now be able to undergo MR procedures following the finalization of a plan by the U.S. Centers for Medicare and Medicaid Services.
The new plan extends coverage for MR scans under certain conditions to patients with implanted pacemakers, implantable cardioverter defibrillators, cardiac resynchronization therapy pacemakers, and cardiac resynchronization therapy defibrillators, for the diagnosis and treatment of diseases or injuries or for improved body part functioning.
“We propose that the evidence is sufficient to conclude that magnetic resonance imaging (MRI) for Medicare beneficiaries with an implanted pacemaker (PM), implantable cardioverter defibrillator (ICD), cardiac resynchronization therapy pacemaker (CRT-P), or cardiac resynchronization therapy defibrillator (CRT-D) is reasonable and necessary for the diagnosis or treatment of illness or injury or to improve the functioning of a malformed body member under section 1862(a)(1)(A) of the Social Security Act under certain circumstances,” said CMS in a
summary of the plan.



Ad Statistics
Times Displayed: 346433
Times Visited: 21062 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
The presence of these devices originally prevented patients from undergoing MR scans unless they were performed within clinical studies, in accordance with the coverage evidence development requirement of the Medicare National Coverage Determinations (NCD) Manual.
The new plan,
proposed in January, eliminates this requirement along with two ideas under consideration in the proposal to mandate for patients to wait six weeks following implants or surgical modifications before undergoing an MR exam, and that they not be dependent on pacemakers.
Coverage is specifically extended to those with FDA-approved pacemakers, implantable cardioverter defibrillators, cardiac resynchronization therapy pacemakers, and cardiac resynchronization therapy defibrillators.
Those with devices that lack FDA-labeling for MR may also undergo scans provided the MR field strength is 1.5 Tesla using normal operating mode and the implant has no fractured, epicardial, or abandoned leads.
In addition, the facility must implement a checklist that includes an assessment to identify devices; communication of MR benefits and harms to the patient prior to scanning; interrogating and programming devices appropriately based on device and patient characteristics; supervision of exam by a qualified physician, nurse or physician assistant with expertise on implanted devices; observation of patients via visual and voice contact, as well as monitoring of vital signs and cardiac rhythm; the presence of an advanced cardiac life support provider during the scan; and a discharge plan that includes the evaluation of the patient and implant to detect and correct abnormalities developed during MR procedures.
CMS did not respond for comment.
