by
John R. Fischer, Senior Reporter | July 25, 2018
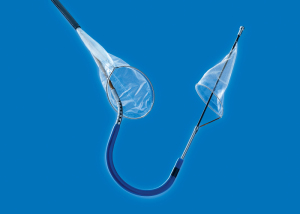
Boston Scientific will take on ownership
of the Sentinel System for stroke
prevention in TAVR patients through
its acquisition of Claret Medical
The Sentinel Cerebral Embolic Protection System, designed to protect patients against stroke during TAVR operations, will soon fall under the ownership of Boston Scientific Corporation following its acquisition of Claret Medical Inc.
Purchasing the private, California-based enterprise for $220 million in cash up-front enables the Massachusetts solution provider to enhance the safety of its product portfolio for TAVR and may potentially open up new avenues of use in procedures for other left heart and endovascular conditions. A potential reimbursement-based milestone payment of up to $50 million is also included as part of the purchase.
“The Sentinel System complements our current adjunctive TAVR offerings which include the Safari2 Guidewire, the iSleeve Expandable Sheath and the Bolt Direct Access Sheath,” Trish Backes, a company spokesperson for Boston Scientific, told HCB News. “While the Sentinel System is indicated for use only with TAVR procedures in the U.S, Boston Scientific will pursue opportunities for the system to be used more broadly as the CE Mark label indicates, in other endovascular procedures like mitral valve repair and replacement, LAAC, and pulmonary vein isolation (PVI) ablation procedures for Afib.”



Ad Statistics
Times Displayed: 6173
Times Visited: 20 Brand-New FDA-cleared Advanced Ultrasound Medical Device available for sale or lease to Wound Care Centers or any other Medical Facilities.The Arobella 1000D is designed for non-contact or debridement ultrasound wound healing therapy, or any other wounds
Cleared in 2017 by the FDA, the Sentinel System captures embolic debris such as calcium or tissue that breaks loose during procedures and is carried through the bloodstream toward the brain where it risks causing neurological and neurocognitive damage including stroke.
Recent studies have found that the onset of clinically-apparent strokes is estimated to take place in approximately four percent of patients within 30 days following a TAVR procedure.
In the pivotal SENTINEL trial, the system reduced such incidences by 63 percent within the first 72 hours following procedures, and when tested in clinical studies, captured debris in 99 percent of TAVR cases, regardless of the type of replacement valve used.
The solution is the only one cleared by the FDA for stroke prevention in patients undergoing TAVR and has been used in nearly 10,000 patients worldwide, a statistic that Boston Scientific plans to build on with the resources at its disposal.
“Boston Scientific has the infrastructure, which includes clinical, manufacturing, sales and distribution channels that will be able to expand patient access to this valuable technology,” said Backes.
Boston Scientific previously
acquired nVision Medical Corporation
as well as Securus Medical Group
and Millipede earlier this year.
Under the agreement, employees of Claret Medical will retain their position under Boston Scientific upon completion of the acquisition.
A closing date for the deal is predicted at some point during the third quarter of 2018 with the agreement subject to customary closing conditions.
The transaction is expected to be irrelevant to adjusted earnings per share in 2018, accretive in 2019 and increasingly accretive thereafter. From a GAAP perspective, it is anticipated to increase less or be possibly more dilutive based on amortization expense and acquisition-related net charges.

