by
Lisa Chamoff, Contributing Reporter | July 01, 2019
From the July 2019 issue of HealthCare Business News magazine
While digital breast tomosynthesis continues to become the standard of care in breast cancer screening and diagnosis, AI technology is emerging to assist with organizing the radiology worklist and with reading the images.
Manufacturers are also investing in more advanced breast MR and breast ultrasound systems.
Here’s a look at what’s new from more than a dozen companies.



Ad Statistics
Times Displayed: 138058
Times Visited: 7972 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
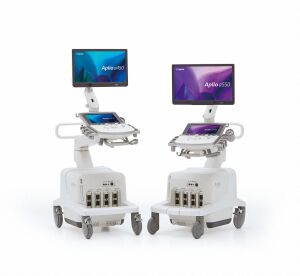
Canon Medical
Canon Medical debuted two new breast ultrasound products at RSNA last year.
The a450 and a550, part of the Aplio a-series of products, both come fully configured with a range of breast transducers. They also feature advanced breast technology such as MicroPure, which enhances the visualization of microcalcifications.
The systems also have ergonomic features, including lighter transducers and thinner cables, that allow the sonographers to scan with less discomfort, said Tina Hodgson, senior manager of solutions marketing for ultrasound at Canon Medical.
“When you sit and scan, you’re twisting your body to see the system and put the transducer on the patient,” Hodgson said. “You want to engage with your patient. The a-series, in particular, has so much flexibility, having the ability of being able to position the monitor and control panel so you’re not in that ‘either/or’ position.”
For interventional procedures, the company introduced a second console, so the sonographer and the doctor performing the biopsy can work together more easily.
In the breast MR space, Canon Medical last year released a 1.5T MR system called the Vantage Orian.
The scanner has the ability to scan the whole body, though for breast MR there is a Sentinelle Breast Coil that can be purchased with the system. The scanner is also available with MSOFT Fat Suppression technology, which helps improve image quality.
Olea Medical, a Canon partner company for image processing and visualization, will introduce a new product for post processing of breast images.
It is pending FDA clearance.
“It is going to be a cutting-edge tool for post processing of breast images and will replace existing post processing technology,” said Suresh Narayan, senior manager for strategic development in MR at Canon Medical. “It will make Canon Medical’s breast imaging portfolio very strong.”
CureMetrix
In March, CureMetrix received FDA Clearance for its cmTriage AI software, which preprocesses and triages radiologists’ mammography worklists.
The software provides a code on the worklist that tells the radiologist whether a reading is suspicious or not suspicious within three to four minutes.