by
Lynn Shapiro, Writer | October 29, 2008
The first electro-magnetic device to be approved for depression has passed FDA muster.
Neuronetics Inc., a privately held company in Malvern, PA, developed the device, called NeuroStar Transcranial Magnetic Stimulation (TMS) Therapy. eTMS therapy involves an electro-magnetic coil that turns on and off, creating a small, electric current in the brain, says Mark George, M.D., Professor of Psychiatry and Neurology at the Medical University of South Carolina (MUSE), who conducted the clinical trials. TMS was also tested on patients at Columbia Medical Center.
Dr. George has been developing the use of TMS since 1993. He tells DOTmed News that unlike Electro Convulsive Therapy (ECT), which produces a seizure in patients so they have to be put to sleep, TMS is not at all painful and requires no anesthesia. With TMS, people can leave their doctor's office without a caretaker after a 40-minute, brain-tuning, typically administered for four to six weeks.




Ad Statistics
Times Displayed: 30213
Times Visited: 748 Stay up to date with the latest training to fix, troubleshoot, and maintain your critical care devices. GE HealthCare offers multiple training formats to empower teams and expand knowledge, saving you time and money
"Rattle Snakes "
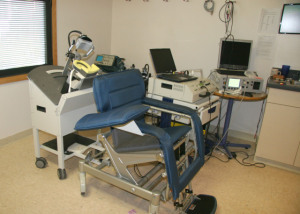
Dr. George says that when patients try TMS, they sit in a comfortable seat that looks like a dentist's chair. The doctor puts the TMS coil over the prefrontal cortex, part of the brain that serves as a rational counterpart to the brain's deep-seeded, highly emotional limbic system, which evolutionarily, was developed long before the prefrontal cortex and often engages in "fight-or-flight behavior.
Dr. George uses a snake analogy to explain the two brain parts: "Say you have a snake phobia and a friend wiggles a snake in front of you, your limbic system would see it as a real snake and you would scream. Your prefrontal cortex would then soothe the fear by recognizing that the snake was only pretend. By stimulating the cortex, we're trying to restore the balance between the cortex and the limbic system and therefore overcome depression," Dr. George says.
"Back in 1993," he adds, "I began my career doing brain imaging. In depressed people, both the prefrontal cortex and the limbic system looked abnormal in brain scans. They were either low functioning or hyper-functioning in depressed and bipolar people."
Efficacy Rate
Dr. George notes that the effects that were seen with TMS in clinical trials--a 15 percent remission rate--were as large as with anti-depressants in the patients tested in his clinical trial, since everyone enrolled in the trial had to have failed to respond to at least one anti-depressant. Patients who failed to get better after trying four or five anti-depressants also got no better after trying TMS, Dr. George says.
"These are people who are looking for new options. And while TMS is not pleasant--it feels like someone is rubbing an eraser on your head--there are no side effects like with pills, which sometimes cause weight gain or cognitive slowing and sexual dysfunction," he says.
Dr. George is Honored
Dr. George was recently honored by NARSAD, the world's leading charity dedicated to mental health research. NARSAD's 21st annual gala, held October 17 at the Waldorf-Astoria in Manhattan, has a 109-member scientific council comprised of leading experts in mental health research. Dr. George received the Falcone Prize for leading the way toward using brain imaging to understand the anatomy of psychiatric disorders.