by
Gus Iversen, Editor in Chief | September 12, 2016
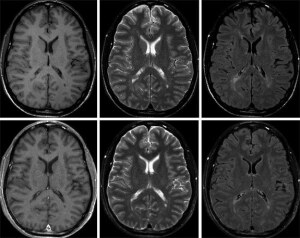
Conventional examination at 1.5T
(top row scan time 14:54 min) compared
to SyMRI examination (bottom row,
scan time 5:48 min). Courtesy: SyntheticMR
GE Healthcare has received FDA clearance for MAGiC, a multi-contrast MR technique developed in partnership with SyntheticMR AB that gives clinicians more data than conventional scans in a fraction of the time.
"MAGiC simultaneously provides a new level of protocol efficiency and tissue characteriztion," said Dr. Howard Rowley, professor of radiology, neurology and neurosurgery at the University of Wisconsin in Madison and president of the American Society of Neuroradiology, in a statement. "The flexibility to post-process a wide variety of key image families with different tissue contrasts from a single acquisition is groundbreaking."
It could also be a major advance for performing faster brain protocols due to the reduced scan time, said Rowley.



Ad Statistics
Times Displayed: 120957
Times Visited: 6963 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
MAGiC is a customized version of SyntheticMR's SyMRI IMAGE software that saves time and money by eliminating the need for additional scans, and lets clinicians manipulate MR images retrospectively. Modifying the image contrast after scanning is not possible with conventional MR imaging.
“FDA clearance of MAGiC is a very important step and a significant achievement by our partner, GE Healthcare." said Stefan Tell, CEO of SyntheticMR. "FDA clearance means that MAGiC now has regulatory approval and is available for sale on most major markets globally.”
Leading up to the approval, GE Healthcare submitted a multi-center blind study to the FDA illustrating that the image quality of MAGiC scans obtained on 1.5T and 3T systems at six facilities for 109 patients were comparable to conventional images — and produced in a fraction of the time.
“This study proves that this technique can improve the way clinicians conduct neuro scans and diagnose their patients," said Eric Stahre, president and CEO, GE Healthcare MR. "Neuro scans are approximately thirty percent of all MR scans; therefore MAGiC could have a tremendous impact on the MR community."
In order to change the image contrast of a MAGiC scan, the user moves the cursor on the interface to change the parameters such as TR, TE and TI. With this flexibility, T1, T2, STIR, T1 FLAIR, T2 FLAIR, Dual IR, Phase Sensitive IR, and Proton Density Weighted images of the brain can all be obtained in a single exam.
This also allows for more advanced tissue analysis, and therefore more precision-based insights into the disease of a particular patient.

