by
John R. Fischer, Senior Reporter | July 17, 2023
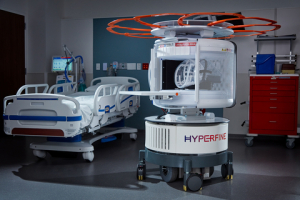
Hyperfine's portable Swoop MR scanner.
Hyperfine has formed an advisory board of stroke experts that will oversee a prospective, international, multisite trial to evaluate the ability of its portable MR scanner, the Swoop system, to identify strokes and viable brain tissues that can be saved.
The point-of-care solution was FDA-cleared in 2020, and users wheel it up to the patient’s bedside to scan them there, increasing workflow and throughput and reducing risks associated with moving and transferring patients in between rooms. Due to its lower cost, it is geared toward acute care facilities, which often lack standard MR scanners.
The study is called ACTION PMR (ACuTe Ischemic strOke detectioN with Portable MR), and will be led by Dr. W. Taylor Kimberly of Massachusetts General Hospital, Dr. Adnan Siddiqui of the University at Buffalo, Dr. Vivien Lee of the Ohio State University Wexner Medical Center, and Dr. Keith Muir of the University of Glasgow.



Ad Statistics
Times Displayed: 344592
Times Visited: 21056 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
“Refining stroke diagnosis and care is central to all we do; as such, our research will focus on patients who don’t immediately qualify for thrombectomy,” said Kimberly, chief of the division of neurocritical care at MGH, in a statement.
Kimberly and his colleagues will also evaluate and further develop the potential of the Swoop system for stroke care. In previous studies conducted at Yale University, the system has shown promise in identifying different types of strokes, including intracranial hemorrhages.
The company recently
nabbed FDA clearance for Swoop’s latest AI-based software update, which improves image quality in diffusion-weighted imaging sequences by increasing signal-to-noise ratio; and increases field-of-view by 10% for T1, T2, and FLAIR sequences, to match previously improved DWI sequences’ 20 cm field of view, and for more clearly visualizing brain pathology.
In addition to the U.S., the system is approved for use in Canada, Australia, the U.K., the EU, and New Zealand.
