by
Brendon Nafziger, DOTmed News Associate Editor | October 06, 2011
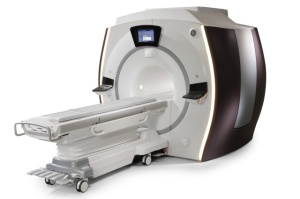
Discovery MR750w (Credit:GE)
GE Healthcare's new wide-bore 3-Tesla MRI, with a suite of radiofrequency coils and "accent lighting" designed to make patients more comfortable, received Food and Drug Administration 510(k) clearance, the Waukesha, Wis.-based company said Thursday.
The Discovery MR750w features a wide, 70-centimeter bore to accommodate obese, elderly, claustrophobic and young patients, GE said. It also has a 50-centimeter field of view for faster scans and OpticalRF technology to minimize signal degradation during analog-to-digital conversion.
"Under that sleek exterior is some of the most advanced technology we've ever built into an MR," Jacques Coumans, general manager of premium MR with GE, said in a statement. "Our goal has been to deliver a 3-T system with a 70-centimeter bore with no compromises."



Ad Statistics
Times Displayed: 348171
Times Visited: 21065 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
To make scans more pleasant for patients, the device features mellow LED accent lights on its sides and the Geometry Embracing Method, or GEM, suite of coils and patient positioning table, which allow for feet-first imaging on all exams and which follow the natural contours of the body for greater patient comfort, GE said.
For the operator, the unit comes with an in-room control console and the "IntelliTouch" patient positioning system, for faster set-up times, GE said.
The Discovery MR750w made its debut at the Radiological Society of North America's annual meeting last November.
