by
John R. Fischer, Senior Reporter | June 07, 2023
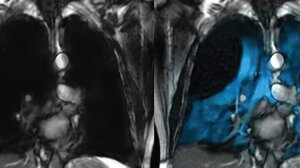
Philips has combined its MR 7700 scanner with Polarean's XENOVIEW inhalable contrast agent for assessing lung ventilation on MR scans.
Philips has partnered with Polarean to bring ZENOVIEW, the first and only inhaled MR contrast agent, to its MR 7700 system, enabling users to directly image lung function with MR scans.
CT imaging is often the primary imaging tool for anatomically evaluating lungs, but XENOVIEW makes it possible to do this on MR scanners, expanding their use as a quantitative tool into pulmonary medicine, and sparing patients from CT scans’ ionizing radiation and associated risks. The hyperpolarized MR contrast agent can be used in adults and pediatric patients, ages 12 and up, in all lung procedures involving interventions, such as pulmonary valve and lung surgery.
Philips’ MR 7700 and XENOVIEW were recently used together for the first time in North America at Cincinnati Children’s Hospital Medical Center, and showcased at the 2023 International Society for Magnetic Resonance in Medicine Annual Meeting and Exhibition in Toronto, Canada.



Ad Statistics
Times Displayed: 344592
Times Visited: 21056 MIT labs, experts in Multi-Vendor component level repair of: MRI Coils, RF amplifiers, Gradient Amplifiers Contrast Media Injectors. System repairs, sub-assembly repairs, component level repairs, refurbish/calibrate. info@mitlabsusa.com/+1 (305) 470-8013
Polarean uses hyperpolarization physics to transfer the energy in a circulatory polarized laser beam to Xenon nuclei to create hyperpolarized Xenon gas, enhancing its MR signal by 10,000 fold to visualize lung ventilation. While traditional CT scans can only image the first few branches of lung airways, Xenon travels with the oxygen down to the small airways, enabling visualization of gas distribution.
“Imaging lung ventilation is more sensitive and repeatable than conventional pulmonary function tests. In addition, this technology provides regional information about lung function. These characteristics make it possible to detect pulmonary diseases earlier and to better evaluate the therapy,” Ruud Zwerink, general manager of MR at Philips, told HCB News.
Philips
received FDA clearance for the 7700 3T MR system in May of 2022. The scanner is built with powerful XP gradients for improved high-quality diffusion imaging.

