by
Lisa Chamoff, Contributing Reporter | April 26, 2021
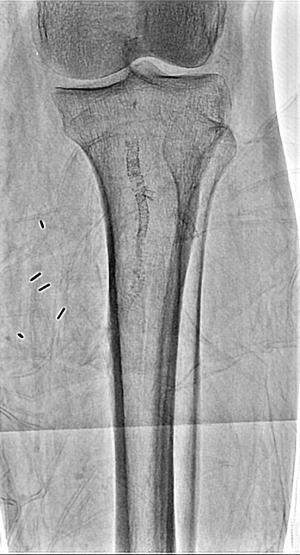
Canon Medical SKY+ 12 inch FOV which is standard FOV
From the April 2021 issue of HealthCare Business News magazine
Manufacturers of fixed and mobile C-arms continue to seek out the holy grail of radiology – providing the clearest images at the lowest possible dose. Several companies are achieving this with the use of high-definition and 3D imaging, coupled with artificial intelligence (AI) to decrease radiation exposure.
At the same time, they’re looking to make C-arms more mobile than ever with intuitive touch-screen user interfaces.
Here’s a look at what's new in the C-arm space from several manufacturers.




Ad Statistics
Times Displayed: 101
Times Visited: 0 Stay up to date with the latest training to fix, troubleshoot, and maintain your critical care devices. GE HealthCare offers multiple training formats to empower teams and expand knowledge, saving you time and money.
Canon Medical Systems
At last year’s RSNA annual meeting, Canon Medical Systems announced that it was making its 12-inch-by-16-inch True Hi-Def Detector available on the Alphenix Sky+, a ceiling-mounted fixed C-arm and 4D CT, combined CT/angiography room. The technology was previously available on the Alphenix Core+ and Alphenix Biplane, which were released two years ago. The technology allows for magnification with 76 micron pixel resolution at 3 inches, 2.3 inches and 1.5 inches.
“This expands the applications for this technology,” said Kevin Maguire, managing director of the vascular business unit at Canon Medical Systems USA. “We believe the high definition technology may be very beneficial in supporting challenging interventions” across a variety of interventional radiology procedures.
Canon Medical SKY+Hi-Def 3 inch FOV now the stent fracture can be seen
A study of over 6,300 neurointerventional procedures comparing the use of high definition technology to standard imaging technology on Canon Medical’s 12-inch-by-12-inch detector quantified a decrease in both cumulative dose and total procedure time with the use of high definition imaging, according to Maguire.
ControlRad
In September 2020, ControlRad announced FDA clearance for use of the ControlRad Trace solution, designed to reduce radiation exposure on the GE OEC 9900 Elite, one of the most commonly used mobile C-arm models in the U.S.
