by
John R. Fischer, Senior Reporter | July 09, 2020
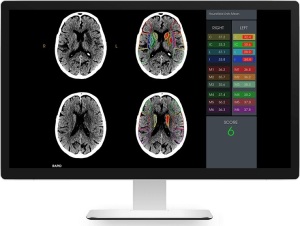
Rapid ASPECTS is the first neuroimaging analysis system in the computer-assisted diagnostic software (CADx) category.
The FDA has given RapidAI the green light to proceed with the release of Rapid ASPECTS, the first neuroimaging analysis system in the computer-assisted diagnostic software (CADx) category.
The solution is the only neuroimaging device capable of improving physician’s interpretations of strokes from non-contrast CT scans using a standardized ASPECT score and determination of whether a patient is eligible to undergo thrombectomy.
“For the first time there is a software that allows MDs to improve their accuracy for reading CT scans of the brain in stroke patients,” Dr. Greg Albers, professor of neurology at Stanford University, director of the Stanford Stroke Center and co-founder of RapidAI, told HCB News. “This is especially significant for less experienced readers.”



Ad Statistics
Times Displayed: 6044
Times Visited: 20 Brand-New FDA-cleared Advanced Ultrasound Medical Device available for sale or lease to Wound Care Centers or any other Medical Facilities.The Arobella 1000D is designed for non-contact or debridement ultrasound wound healing therapy, or any other wounds
Rapid ASPECTS is based on the Alberta Stroke Program Early CT Scoring (ASPECT) system, and is equipped with a validated machine-learning algorithm to automatically detect the ASPECTS regions of the brain. It then generates an ASPECT score to indicate early signs of brain infarction on NCCT scans, which helps physicians identify areas of irreversible brain injury and determine if a patient is eligible for thrombectomy.
The technology automates and standardizes workflow by helping stroke teams across hospital sites and referral networks quickly determine if a patient is eligible for thrombectomy, enabling them to triage faster and make quicker transfer decisions. As timing in stroke assessment is crucial, the ability to act faster with the system is expected to lead to better patient outcomes.
“Signs of a stroke can be very subtle on a CT scan during the first several hours after symptom onset. The software has been shown to improve physicians' accuracy for detecting these subtle signs,” said Albers, adding that the solution will be a strong asset for “neurologists, emergency medicine specialists, radiologists and neurosurgeons.”
Rapid ASPECTS is part of the AI-based Rapid platform that produces advanced images from non-contrast CT, CT angiography, CT perfusion, and MR diffusion and perfusion scans. Others include Rapid ICH, Rapid ASPECTS, Rapid CTA, Rapid CTP, and Rapid MRI.
The solution is available internationally and will be on sale in the U.S. starting later this month.

